Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
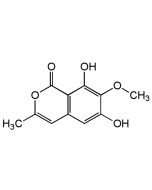
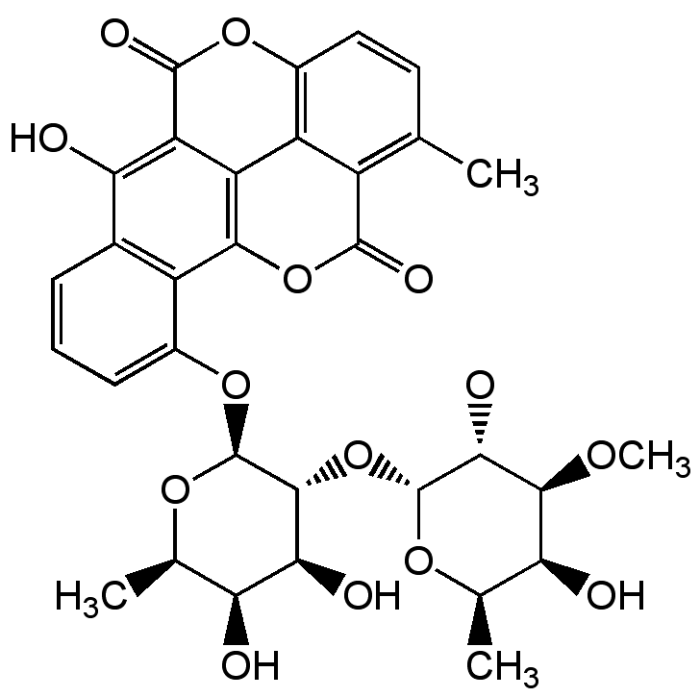
Chartreusin
As low as
155
CHF
CHF 155.00
In stock
Only %1 left
BVT-0005-M0055 mgCHF 155.00
| Product Details | |
|---|---|
| Synonyms | Lambdamycin |
| Product Type | Chemical |
| Properties | |
| Formula |
C32H32O14 |
| MW | 640.6 |
| Merck Index | 14: 2045 |
| CAS | 6377-18-0 |
| RTECS | FL7350000 |
| Source/Host Chemicals | Isolated from Streptomyces chartreusis. |
| Purity Chemicals | ≥98% (HPLC) |
| Appearance | Yellow solid. |
| Solubility | Soluble in acetone; insoluble in water. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | PONPPNYZKHNPKZ-CIFTUGPDSA-N |
| Smiles | CO[C@H]1[C@@H](O)[C@@H](C)O[C@H](O[C@@H]2[C@@H](O)[C@@H](O)[C@@H](C)O[C@H]2OC2=CC=CC3=C2C2=C4C(C(=O)OC5=C4C(=C(C)C=C5)C(=O)O2)=C3O)[C@@H]1O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | +4°C |
| Use/Stability |
Stable for at least 1 year after receipt when stored at +4°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Antibiotic
- Antitumor compound.
- Topoisomerase II inhibitor.
- Induces single-strand scission in DNA in the presence of reducing agents.
- Apoptosis inducer.
- Inhibitor of glioblastoma multiforme (GBM).
Product References
- Chartreusin, a new antibiotic produced by Streptomyces chartreusis, a new species: B. E. Leach, et al.; J. Am. Chem. Soc. 75, 4011 (1953)
- Chartreusin, a glycosidic antitumour antibiotic from Streptomyces: J.A. Beisler; Prog. Med. Chem. 19, 247 (1982) (Review)
- Biochemical characterisation of elsamicin and other coumarin-related antitumour agents as potent inhibitors of human topoisomerase II: A. Lorico & B.H. Long; Eur. J. Cancer 14, 1985 (1993)
- Chartreusin, elsamicin A and related anti-cancer antibiotics: J. Portugal; Curr. Med. Chem. Anticancer Agents 3, 411 (2003) (Review)
- Biosynthesis of the antitumor agent chartreusin involves the oxidative rearrangement of an anthracyclic polyketide: Z. Xu, et al.; Chem. Biol. 12, 579 (2005)
- In silico studies on marine actinomycetes as potential inhibitors for glioblastoma multiforme: P. Kirubakaran, et al.; Bioinformation 6, 100 (2011)
- Synthetic Remodeling of the chartreusin pathway to tune antiproliferative and antibacterial activities: N. Ueberschaar, et al.; JACS 135, 17408 (2013)
- Rational design of an apoptosis-inducing photoreactive DNA intercalator: N. Ueberschaar, et al.; Angew. Chem. Int. Edit. 52, 6185 (2013)