Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
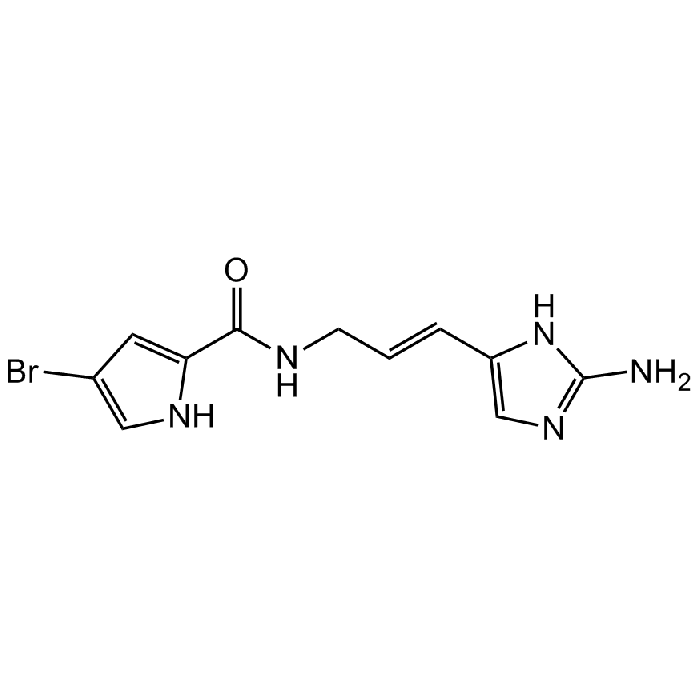
Hymenidin
As low as
230
CHF
CHF 230.00
In stock
Only %1 left
AG-CN2-0503-M0011 mgCHF 230.00
| Product Details | |
|---|---|
| Synonyms | N-[(2E)-3-(2-Amino-1H-imidazol-5-yl)prop-2-en-1-yl]-4-bromo-1H-pyrrole-2-carboxamide; Hymenidine; 2-Debromooroidin |
| Product Type | Chemical |
| Properties | |
| Formula |
C11H12BrN5O . H2O |
| MW | 310.2 . 18.0 |
| CAS | 107019-95-4 |
| Source/Host Chemicals | Isolated from sponge Stylissa sp. |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Amorphous solid. |
| Solubility | Soluble in DMSO, ethanol or methanol. |
| Identity | Determined by 1H-NMR and MS. |
| InChi Key | KHJREOQCERRAME-OWOJBTEDSA-N |
| Smiles | O=C(NC/C=C/C1=CN=C(N)N1)C2=CC(Br)=CN2 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Inhibitor of CDK5/p25 (IC50=4µM) and GSK-3β (IC50=12µM). Potentially useful in neuronal diseases research.
- Antagonist of serotonergic receptors.
- Shown to reduce voltage-dependent calcium elevation.
- Moderate anticancer compound. Inhibits cell growth in a panel of cancer cell lines at low μM range.
- Antiprotozoal agent.
- Antibacterial. Plays a role in the sponge antibacterial defense.
Product References
- A novel antagonist of serotonergic receptors, hymenidin, isolated from the Okinawan marine sponge Hymeniacidon sp: J. Kobayashi, et al.; Experientia 42, 1176 (1986)
- Inhibition of cyclin-dependent kinases, GSK-3beta and CK1 by hymenialdisine, a marine sponge constituent: L. Meijer, et al.; Chem. Biol. 7, 51 (2000)
- Brominated pyrrole alkaloids from marine Agelas sponges reduce depolarization-induced cellular calcium elevation: U. Bickmeyer, et al.; Toxicon 44, 45 (2004)
- Marine compounds for the therapeutic treatment of neurological disorders: D. Alonso, et al.; Expert Opin. Ther. Pat. 15, 1377 (2005)
- Antineoplastic agents 470. Absolute configuration of the marine sponge bromopyrrole agelastatin A: G.R. Pettit, et al.; Oncol. Res. 15, 11 (2005)
- Bromopyrrole alkaloids as lead compounds against protozoan parasites: F. Scala, et al.; Mar. Drugs 8, 2162 (2010)
- Chemical defense against predators and bacterial fouling in the Mediterranean sponges Axinella polypoides and A. verrucosa: M. Haber, et al.; MEPS 422, 113 (2011)







