Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
Asperphenamate
As low as
110
CHF
CHF 110.00
In stock
Only %1 left
AG-CN2-0171-C250250 µgCHF 110.00
AG-CN2-0171-M0011 mgCHF 330.00
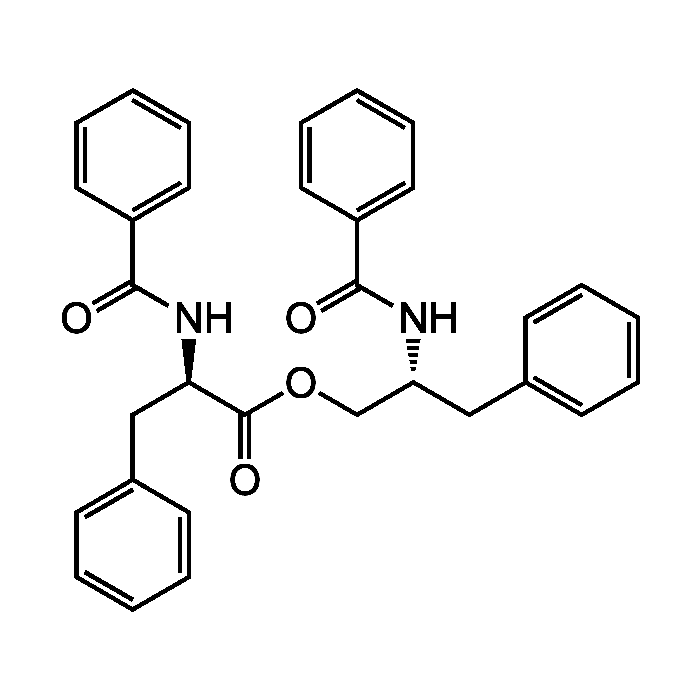
| Product Details | |
|---|---|
| Synonyms | Anabellamide; Asjanin; Auranamide; NSC 306231; N-Benzoyl-phenylalanine-2-benzoylamino-3-phenylpropyl ester |
| Product Type | Chemical |
| Properties | |
| Formula |
C32H30N2O4 |
| MW | 506.6 |
| CAS | 63631-36-7 |
| Source/Host Chemicals | Isolated from Aspergillus sp. |
| Purity Chemicals | ≥95% (HPLC) |
| Appearance | White solid. |
| Solubility | Soluble in ethanol, methanol or DMSO. |
| InChi Key | CVULDJMCSSACEO-FQLXRVMXSA-N |
| Smiles | O=C(OC[C@@H](CC1=CC=CC=C1)NC(=O)C1=CC=CC=C1)[C@@H](CC1=CC=CC=C1)NC(=O)C1=CC=CC=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Anticancer compound.
- Cytotoxic against human breast cancer cells.
- Induces autophagic cell death in MCF-7 cells.
- Moderate radical scavenger.
- Weak acetylcholinesterase (AChE) inhibitor.
- Shows moderate trypanocidal activity.
Product References
- Two metabolites from Aspergillus flavipes: A.M. Clark, et al.; Lloyda 40, 146 (1977)
- Cytotoxic and anti-HIV principles from the rhizomes of Begonia nantoensis: P.W. Wu, et al.; Chem. Pharm. Bull. 52, 345 (2004)
- A new method for asperphenamate synthesis and its antimicrobial activity evaluation: A.M. Pomini, et al.; Nat. Prod. Res. 20, 537 (2006)
- Total synthesis and anticancer activity studies of the stereoisomers of asperphenamate and patriscabratine: L. Yuane, et al.; Chin. Chem. Lett. 21, 155 (2010)
- JNK-dependent Atg4 upregulation mediates asperphenamate derivative BBP-induced autophagy in MCF-7 cells: Y. Li, et al.; Toxicol. Appl. Pharmacol. 263, 21 (2012)
- Two new Penicillium species Penicillium buchwaldii and Penicillium spathulatum, producing the anticancer compound asperphenamate: J.C. Frisvad, et al.; FEMS Microbiol. Lett. 339, 77 (2013)
- In vitro acetylcholinesterase activity of peptide derivatives isolated from two species of Leguminosae: C.Q. Alves, et al.; Pharm. Biol. 51, 936 (2013)
- Antioxidant activity of compounds isolated from the root woods of Erythrina droogmansiana: A.J.G. Yaya, et al.; Int. J. Pharm. Sci. Drug Res. 6, 160 (2014)
- Two trypanocidal dipeptides from the roots of Zapoteca portoricensis (Fabaceae): N.J. Nwodo, et al.; Molecules 19, 5470 (2014)






